 It's summer in the Northern Hemisphere, and in most places that means mosquitos are out for our blood, which wouldn't be much to give up if it weren't for the itchy -- and in many parts of the world, deadly -- package that the tiny insect leaves behind. Plasmodium falciparum is the human malaria pathogen that kills over a million people annually around the world (largely infants, young children and pregnant women, most of them in Africa). One approach to combatting the spread of the disease is to genetically engineer a mosquito that cannot transmit the parasite and yet is able to reproduce with mosquitos that do, in order to infiltrate and alter the population overall to become one that is benign (except for the itching).
It's summer in the Northern Hemisphere, and in most places that means mosquitos are out for our blood, which wouldn't be much to give up if it weren't for the itchy -- and in many parts of the world, deadly -- package that the tiny insect leaves behind. Plasmodium falciparum is the human malaria pathogen that kills over a million people annually around the world (largely infants, young children and pregnant women, most of them in Africa). One approach to combatting the spread of the disease is to genetically engineer a mosquito that cannot transmit the parasite and yet is able to reproduce with mosquitos that do, in order to infiltrate and alter the population overall to become one that is benign (except for the itching).
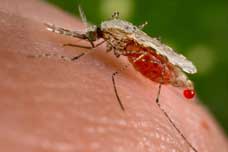
[The Anopheles stephensi mosquito, right, is a major transmitter of malaria in India and the Middle East. Photo by Jim Gathany / Centers for Disease Control & Prevention]
Some of the most exciting advances in the development of these transgenic mosquitos are taking place at the University of California Irvine, in the research lab of Anthony A. James, Professor of Molecular Biology and Biochemistry. In a recent paper published in PNAS, Transgenic Anopheles stephensi coexpressing single-chain antibodies resist Plasmodium falciparum development, James and his colleagues at UCI and the Pasteur Institute in Paris state the problem this way:
Insecticide and parasite drug resistance continue to hinder malaria eradication efforts and innovative approaches to disease control are needed to complement traditional methods.
The team's success James describes succinctly in a UCI interview:
"This is the first model of a malaria vector with a genetic modification that can potentially exist in wild populations and be transferred through generations without affecting their fitness.”
[An earlier video exploring the mosquito research in Dr. James' lab]
The Irvine lab research began with a mouse model. After injecting the mice with the human form of malaria, Plasmodium falciparum, they found that the mice naturally produced antibodies to kill the parasite. Working from there, James' team exploited the molecular components of this mouse immune-system response and engineered genes that could produce the same response in mosquitoes. The key was to do this without compromising the fitness of the mosquitos themselves, so that they could reproduce and spread the genetic modification, which is essentially the disruption of an amino acid chain.
Better yet, the modification that James' research lab has managed to engineer in the Anopheles stephensi mosquito is transferable to other varieties of mosquito that harbor other dangerous diseases like dengue fever, or malaria in another part of the world, such as Africa. They may even be useful in the case of an outbreak of West Nile virus in more temperate climates.
In May we reported on another, very different effort to reduce deaths by malaria by a Southern California research lab. That piece, UCLA Research Wizards Use Gaming to Diagnose Malaria in Telepathology Project, looked at the work of Adogan Ozcan at UCLA and took as its task the more accurate and accessible diagnosis of the disease in small clinics and remote hospitals.

Biotechnology Calendar, Inc. will be holding its 12th annual Irvine BioResearch Product Faire event on the UCI campus October 2, 2012. This professional show is an excellent opportunity for life scientists and lab equipment specialists to come together and discuss lab technologies to make every lab run at maximum efficiency. The UC Irvine BRPF event is one of three tradeshows held over a three-day period in the greater Los Angeles area:
- 10/02/2012 -- 12th Annual BRPF event, UC Irvine
- 10/03/2012 -- 7th Semiannual Front Line event, University of Southern California, Health Sciences Campus
- 10/04/2012 -- 29th Semiannual BVS event, UCLA
For more information on exhibiting at UC Irvine, click the button below. Explore our website and see the complete 2012 Show Schedule, then give one of our sales associates a call.


