(Image courtesy of Wikimedia Commons)
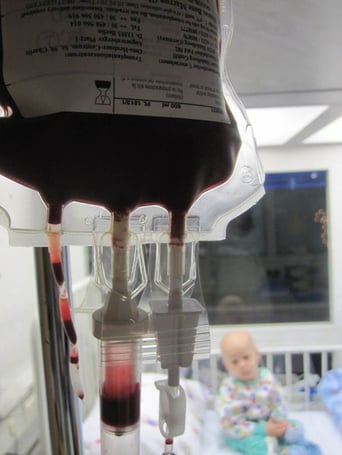
Finding matching donors for patients who need bone marrow transplants can be challenging. For ethnic minorities the chances of finding an unrelated donor who is a match through the national donor registry is less than 35 percent. However, thanks to a three year, $600,000 grant from the Leukemia and Lymphoma Society researchers and physicians at the University of Arizona-Tucson are hoping to improve the odds of survival for those patients. This new research funding will be used to support ongoing biomedical research at the UA Steele Children’s Research Center into improving haploidentical bone marrow transplantations (haplo-BMT) as well as launching clinical trials at the UA Cancer Center.
How is haplo-BMT different from traditional bone marrow transplants?
Haploidentical bone marrow transplants could be life-saving for patients who need a transplant when a related or unrelated donor match cannot be found. Haplo-BTM provides an alternative source of stem cells, which means the donor does not have to be a perfect match. He or she can be a “half-match” and still be a suitable donor, thus allowing a patient’s parent, child, or sibling to donate. The University of Arizona has already successfully treated 12 patients with this groundbreaking procedure at its Banner-University Medical Center Tucson and Banner Children’s at Diamond Children’s Medical Center.
However, according to Emmanuel Katsanis, MD, Professor of Pediatrics, Medicine, Pathology and Immunobiology, and Director of the UA Cancer Center Blood and Marrow Transplant Program, the research conducted by him and his team at the UA Steele Center has “shown that we can improve upon what’s currently the standard of care, in regard to haplo-BMT treatment.” Their research showed that replacing the drug Cyclophosphamide with the novel drug Bendamustine after haplo-BMT improved survival rates in mice. It does this by reducing graft versus host disease, while at the same time increasing the graft versus leukemia effect.
When will clinical trials be held?
According to the UA news site, Dr. Katsanis has written the phase I/II protocols for clinical trials based on his teams findings. He says with this funding, “…we now can move our research from ‘bench to bedside.’ We hope to start the clinical trial this fall.” He also points out that “Haplo-BMT is particularly important in Arizona where we have a large population of Hispanics, who have a very low chance of finding an unrelated bone marrow match through the national registry.”
The director of the UA Steele Children’s Research Center, Fayez K. Ghishan, MD, adds, “This grant enables us to advance our basic science cancer research that ultimately will lead to improved outcomes for patients receiving haplo-BMT.”
About Dr. Katsanis and the University of Arizona Tucson:
Dr. Katsanis is a Professor of Pediatrics, Medicine, Immunobiology, Pathology and a member of the University of Arizona Cancer Center, Steele Children’s Research Center and the Bio5 Institute. He is also the Chair of Pediatric Cancer Research and the Head of the Division of Pediatric Hematology/Oncology/BMT. He joined the faculty at UA in 1997. As a physician-scientist, Dr. Katsanis provides clinical care for children with cancer and blood disorders at Diamond Children’s at University Medical Center and conducts research at the UA Steele Children's Research Center.
The University of Arizona, Tucson is home to the Steele Children’s Research Center, the state’s only academic pediatric research center designated by the Arizona Board of Regents. The physician-scientists there provide care to hospitalized patients at UA’s Banner Children’s at Diamond Children’s Medical Center. Laboratory equipment suppliers interested in meeting face to face with the many researchers at University of Arizona should plan to attend the 14th Annual BioResearch Product Faire™ this November. This premier tradeshow is hosted by Biotechnology Calendar, Inc. For more information call (530) 272-6675 or click on the link below. Researchers attend for free!




